Understanding Drug Labels
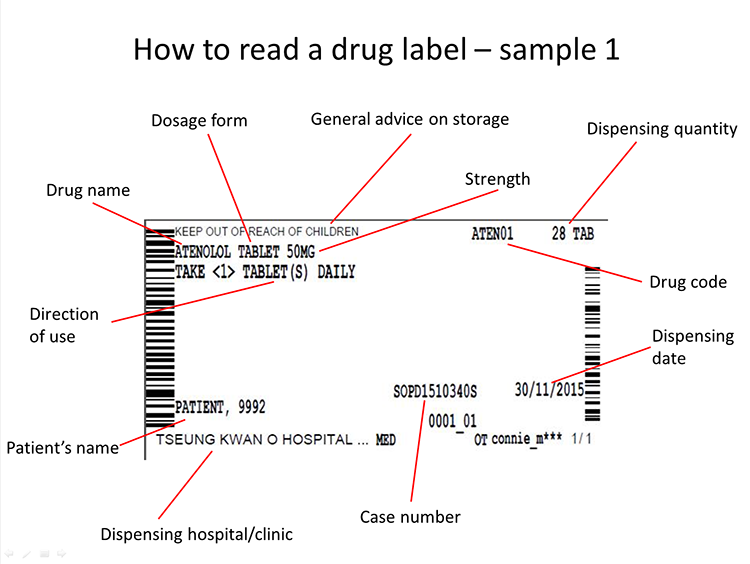
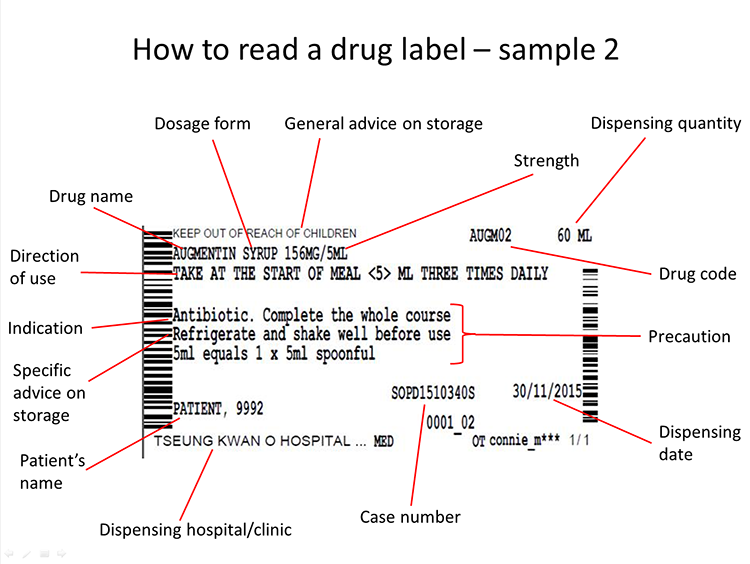
Use drugs in accordance with the instructions on the label, especially the below points:
-
The number of pills for each dose is usually whole number (e.g. 1, 2, 3...), while certain pills may be taken in partial portion like 1/4 or 1/2
5 ml equals 1 spoonful
-
Daily dosage: usually the drugs are to be taken 1 to 4 times per day in 4 to 12-hour intervals; certain drugs are to be taken every other day or every odd/even day
Medication time: morning / evening / before sleep
when necessary
Using Pill Organizer
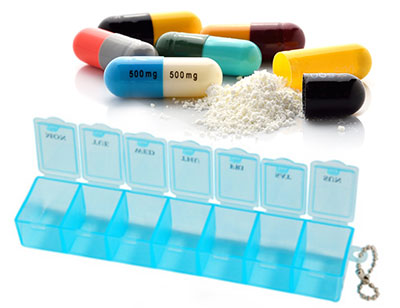
Advantages
- Useful for planning medication order and time and preventing overdose for the elderly patient
- Allows to monitor whether the elderly patient have taken the correct dosage
- Maximum daily dose : e.g. Paracetamol 500mg
Maximum daily dose 8 pills = The patient should take no more than 8 pills in 24 hours since the first dose
- Drugs should be stored in a cool, dry place, but not the refrigerator unless specified
- Refrigerating drugs may affect their stability and effectiveness except for designated ones, e.g. injections, vaccines and eye drops to be refrigerated at 2-8°C
- Store drugs in safe places inaccessible to young children
- Do not mix other drugs in the same container to avoid confusion and misuse
- Drugs should be stored in containers with proper labels, and in bags with proper labels after dose, so medical personnel and family members can look into your medication condition if necessary
- During follow-up or emergency appointments, bring along the drugs and corresponding labels for medical personnel's reference
- Only split a pill into the portion you need to take at the moment instead of future dosage -- exposed drugs degrade

The best way to split a pill in half is to use a pill splitter, which can handle pills in various shapes with correct usage
Remember the following when splitting a pill:
- Place the pill on the triangular base against both sides to avoid uneven cut
- Pull the cover down slowly until the blade touches the pill, then press it down to split the pill
- Regularly clean the splitter to remove pill residue that may rust the blade
Refer to this video on using pill splitter
- Do not take improperly stored drugs that are expired, degraded (discoloured, smelling or stale) or without clear labels
- Eye drops and ear drops can be stored until the expiry date on the label before opening; after opening, it expires in the conditional expiry period on the label (which is normally 1 month)
- Eye drops and ear drops without preservatives lasts only for 12 to 24 hours, while some should be disposed right upon opening and use
- Eye drops and ear drops with preservatives normally expires in 30 days; a few with special ingredients can last for up to 180 days
- Expired or unused drugs after treatment period can be disposed as common domestic waste
FAQ
Q: Should drugs be taken before or after meals?
A: If this is not specified on the label, the common principle is to take the drug before meals and continue if it does not cause stomach discomfort; patients with stomach disease (e.g. ulcer and indigestion, etc.) should take drugs after meals if the aforementioned attempt yields opposite result
Medication instructions are usually stated clearly for drugs that must be taken before meals (e.g. Ziprasidone, aspirin and metformin) or after meals (e.g. e.g. Alendronate, chloestyramine and penicillin)
Q: What is the difference between CR and SR tablets?
A: CR = Controlled release tablets with engineered pharmacokinetics that changes the release and absorption
SR = Sustained release tablets with engineered pharmacokinetics that prolongs the release and absorption
Under circumstances, CR and SR tablets are more effective in maintaining stable condition for the patient, in contrast with the basic regular release tablets. For example, the CR variants of antihypertensive tablets cut down on high chemical absorption over short period of time, thereby alleviating side effects. By reducing medication frequency, SR tablets lower the risk of patient to miss dose. For instance, SR antibiotics can release chemicals over 24 hours, unlike more common ones that are taken 3 times daily on 8-hour intervals
Q: Can I crush the tablet and mix into milk and soup for patients with difficulties to swallow?
A: This depends on the dosage form. Tablets with CR, SR, ZOK or GITS are of special dosage forms and must not be taken crushed or split; otherwise, it belongs to common dosage form and can usually be taken crushed.
Q: The doctor has been giving me tablets in foil packaging, unlike bottled in the past. Can I unpack a week's dose and store them in the pill organizer? Will this affect the tablets' effect?
A: It is not advised to remove the tablets from their original foil packaging and put them into a pill organizer. The foil packaging protects the tablets from sunlight and moisture and comes with batch number and expiry date printed on it. The tablets should only be unpacked and split into 2 halves right before being taken.
Q: I have a bottle of unopened painkiller that is about to expire. Are expired drugs still safe for use? Will refrigerating prolong storage life?
A: The expiry date is usually indicated on the bottles or grid packaging. Being unopened does not mean its storage life is prolonged. The quality of the drug (including the rate of degradation of active ingredients, toxicity, bacterial content and appearance) is guaranteed only within the producer's suggested expiry date. Using expired drug may lead to the opposite of the desired result.
The common belief that refrigerating drugs can extend storage life is not necessarily true. The storage conditions of the drugs and their active ingredients are all tested by the drug maker in the process of production. Unless specified to be necessary to be stored at 2-8°C, drugs should not be refrigerated and it would otherwise impact the drugs stability. The dehumidifier of the refrigerator may lead to disintegration in tablets, brittleness in capsules and layering plus thickening in liquid medicine.
Related Information